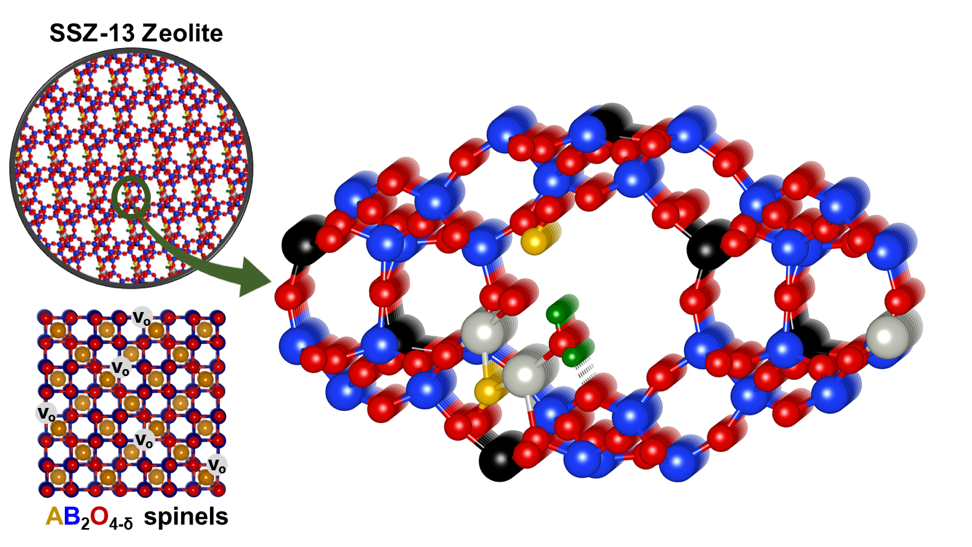
Global environmental concerns have led to stringent vehicle emission (CO, NOx, CH4, light non-methane hydrocarbons) control policies and targets in recent years. While the current technology in practice can convert CO and NOx at high temperatures, during the cold start period (below 200 °C), a major fraction of these components can escape unattended. Thus, to reduce these emissions, sincere efforts are being put to develop better passive NOx adsorbers (PNA) and hydrocarbon traps (HCT). Metal-exchanged zeolites are of particular interest as they exhibit a plethora of dynamically varying active sites for this purpose. These materials are perfect platforms for site-selective multifunctional catalysis owing to zeolite framework heterogeneity. The concept of four-way catalysis not only serves the conversion of CO, NOx and light non-methane hydrocarbons, but also the oxidation of methane to CO2 and H2O. Mixed metal oxides such as spinels are known for their oxygen storage capacity, methane oxidation properties, and stability over a wide temperature range under stoichiometric and oxygen-vacant states. Through density functional theory calculations, our research group is probing intrinsic zeolite active sites and stoichiometric/non-stoichiometric spinel oxides to elucidate their role in these reactions, developing reaction mechanism models and predicting better materials for the future.
Publications:
- Jangjou, Y., Do, Q., Gu, Y., Lim, L.-G., Sun, H., Wang, D., Kumar, A., Li, J., Grabow, L. C. & Epling, W. S. Nature of Cu Active Centers in Cu-SSZ-13 and Their Responses to SO 2 Exposure. ACS Catal. 8, 1325–1337 (2018).
- Song, Y. & Grabow, L. C. Activity Trends for Catalytic CO and NO Co-Oxidation at Low Temperature Diesel Emission Conditions. Ind. Eng. Chem. Res. 57, 12715–12725 (2018).
- Doan, H. A., Sharma, M. K., Epling, W. S. & Grabow, L. C. From Active-Site Models to Real Catalysts: Importance of the Material Gap in the Design of Pd Catalysts for Methane Oxidation. ChemCatChem 9, 1594–1600 (2017).